|
1/21/2024 0 Comments Er lumen phenzymes and mechanoenzymes) and the frequency of catalytic events, there will eventually be a physical limit that determines the ceiling of secretory capacity, namely how much energy to generate and expend as well as how many proteins to synthesize, fold, assemble, package, traffic, and secrete by individual cells in a given time and space. Even if cells expand their capacity by increasing the number of participating catalytic machines ( e.g. Advancements in cell phenotype engineering, metabolic engineering, systems approach, growth medium optimization, and bioreactor technology have all culminated in explosive increases in the production titers of therapeutic human IgGs in recent years ( 16– 19).Īt the cellular level, protein secretion capacity may be thought of as the cumulative efficiency of numerous biochemical reactions and biomechanical steps constituting the biosynthetic secretory pathway. To alleviate such post-translational bottlenecks, various approaches were evaluated to enhance the protein secretion efficiency. Despite the success in boosting protein expression per se through these expression vector engineering approaches, such enhancements did not translate into higher glycoprotein secretion partly because post-translational events such as protein folding/assembly and intracellular vesicular transport steps along the secretory organelles became the new bottlenecks ( 13– 15). Exogenous nucleotide elements also enabled strategies to increase transgene copy number by gene amplification and to suppress epigenetic silencing ( 9– 12). 
To achieve high recombinant protein expression levels in mammalian cells, various cis-acting exogenous nucleotide elements have been engineered into transgene expression cassettes to enhance transcription efficiency, extend message half-life, and increase translation initiation frequency ( 7, 8). Mammalian cell hosts are often preferred for biopharmaceutical production not only just to achieve desired co-translational and post-translational modifications ( 4) but also to exploit the stringent protein quality control mechanisms that only allow the secretion of properly folded and correctly assembled proteins ( 5, 6). Therapeutic human IgGs are often recombinantly produced in variants of CHO cells that were adapted to propagate in suspension culture format. Although immunoglobulins are indispensable as research tools, their potential as human therapeutics has attracted significant interest in recent years in the manufacture of human IgG at large scale ( 2, 3). Immunoglobulins continue to serve as an important model secretory cargo for investigating biochemical processes of oxidative protein folding and subunit assembly in the ER 2 lumen ( 1). Using an in vivo system that reports accumulation of correctly folded IgG, we showed that the ER-to-Golgi transport steps became rate-limiting in cells with high secretory activity. As a result, export-ready IgG accumulated progressively in the ER lumen until a threshold concentration was reached to nucleate crystals. The striking cellular phenotype implicated the efficiency of IgG protein synthesis and oxidative folding exceeded the capacity of ER export machinery. 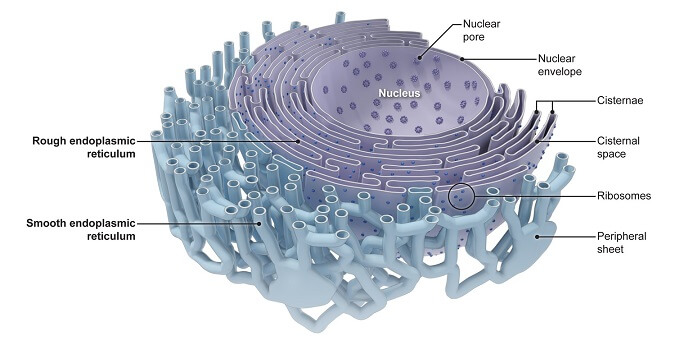
Crystallizing propensity was due to the intrinsic physicochemical properties of the model IgG, and the crystallization was reproduced in vitro by exposing a high concentration of IgG to a near neutral pH. The intra-ER crystals were composed of correctly folded, endoglycosidase H-sensitive IgG. The intra-ER crystal growth was accompanied by cell enlargement and multinucleation and continued until crystals outgrew cell size to breach membrane integrity. Under highly optimized growth conditions, recombinant CHO cells engineered to produce a model human IgG clone started housing rod-shaped crystals in the endoplasmic reticulum (ER) lumen. Increasing protein synthesis until reaching the limit of secretory capacity is one strategy to address this key issue. Although secretory activities may vary in different cell types, what determines the maximum secretory capacity is inherently difficult to study. Protein synthesis and secretion are essential to cellular life.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
